Olivia Isabella posted an update in the group
 Product 2 years, 4 months ago
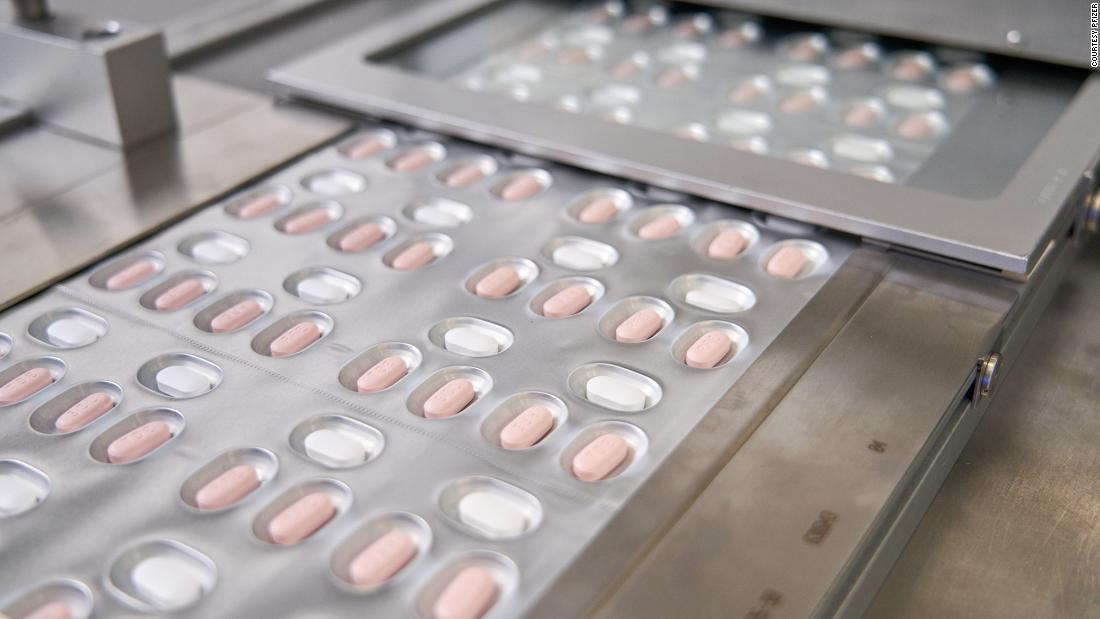
Product 2 years, 4 months agoThe US FDA Has Approved The Second COVID-19 Pill, Called Molnupiravir, From Merck. This Approval Comes A Day After The Agency Approved The First-Ever COVID-19 Pill, Called Paxlovid, From Pfizer. The Pills Are For Mild-To-Moderate COVID-19 Ill People, To Take At Home, Who Are At High-Risk Of Progressing To A Severe Stage Of COVID-19.
Covid pill: FDA authorizes two oral antivirals to treat coronavirus infection – CNNThe US Food and Drug Administration on Thursday authorized Merck’s antiviral pill, molnupiravir, to treat Covid-19 “for the treatment of mild-to-moderate coronavirus disease (COVID-19) in adults with positive results of direct SARS-CoV-2 viral testing, and who are at high risk for progression to severe COVID-19, including hospitalization or death, and for whom alternative COVID-19 treatment options authorized by the FDA are not accessible or clinically appropriate.”